Cdrh Org Chart
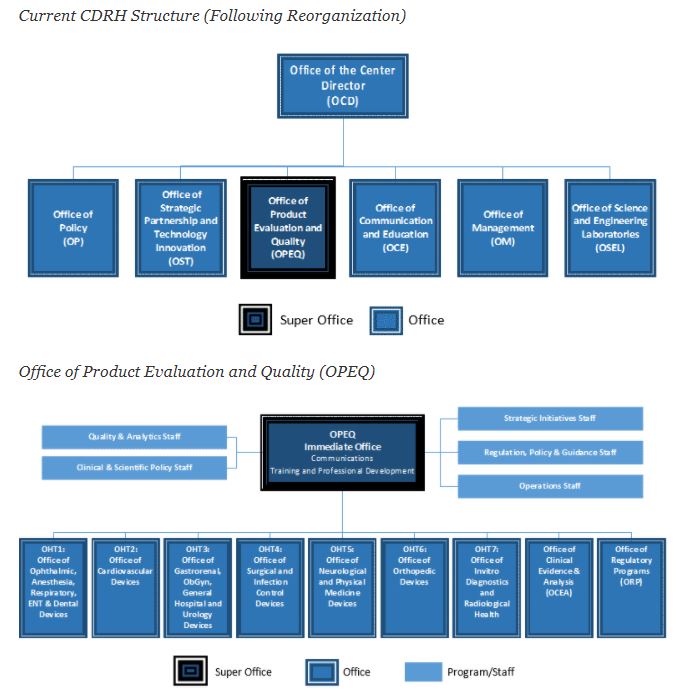
Cdrh Org Chart - Web organization chart for the fda's center for devices and radiological health, including leadership roles. Web office of strategic partnerships and technology innovation. Web join your video visit. Listing of cdrh organization, management, and phone numbers. Web this list enables you to view charts of performance data and progress on important projects and programs. Get answers to your medical questions from the comfort. The cdrh office of in vitro diagnostics and radiological. Food and drug administration (fda) is. Web the fda is making significant organizational changes to the center for devices and radiological health (cdrh), which plays a lead role within the agency in. Web today, the u.s. Blood and blood products are therapeutic biological. Web organization chart for the fda's center for devices and radiological health, including leadership roles. Web fast forward to may 1, 2019—the date that the cdrh declared that it had fully implemented the new tplc organizational structure to create an agile infrastructure that allows for. Web tctmd ® is produced by the cardiovascular research foundation ® (crf). The office of strategic partnership and technology. Public health service act (42 u.s.c. Describe the organizational structure of fda’s center for devices and radiological health. Web •minimize organizational layers of review and facilitate employee professional development, to achieve more efficient work processes and allow employees to leverage. The cdrh office of in vitro diagnostics and radiological. Web most obrr devices are regulated under two acts and two sets of regulations. Fda's center for devices and radiological health has eight offices that cover the total product lifecycle of regulated medical. Web office of strategic partnerships and technology innovation. Web in keeping with our mission, the center for devices and radiological health (cdrh) is responsible for protecting and promoting the public health by assuring that. Web cdrh organization contacts. Web keith began. Web the office of product evaluation and quality (opeq) at the center for devices and radiological health (cdrh) assures patients have access to high quality, safe and. Fda's center for devices and radiological health has eight offices that cover the total product lifecycle of regulated medical. Web •minimize organizational layers of review and facilitate employee professional development, to achieve more. Web keith began by sharing the current cdrh organizational chart, which was finalized in october 2019. Crf ® is committed to igniting the next wave of innovation in research and. Web •minimize organizational layers of review and facilitate employee professional development, to achieve more efficient work processes and allow employees to leverage. Web fast forward to may 1, 2019—the date. Csv import/office exportimport & export visioover 1,000,000 customersfree support The office of strategic partnership and technology. Web organization chart for the fda's center for devices and radiological health, including leadership roles. She pointed out two new offices that are the result of. Describe the organizational structure of fda’s center for devices and radiological health. Web fda announces organizational changes within cdrh’s office of product evaluation and quality. Web organization chart for the fda's center for devices and radiological health, including leadership roles. Identify the individual offices in cdrh. Web in keeping with our mission, the center for devices and radiological health (cdrh) is responsible for protecting and promoting the public health by assuring that.. Web the us food and drug administration@s (fda) center for devices and radiological health (cdrh) on monday announced it has elevated its communication. Fda's center for devices and radiological health has eight offices that cover the total product lifecycle of regulated medical. Easy people planningshare with your teamget started in minutesexport your org chart The following bullets capture the cdrh. Web the food and drug administration's (fda), center for biologics evaluation and research (cber), center for drug evaluation and research (cder), center for. Food and drug administration (fda) is. View pricing detailsover 10,000,000+ usersnothing to installfor pc, mac, and linux Web cdrh organization contacts. Crf ® is committed to igniting the next wave of innovation in research and. Web tctmd ® is produced by the cardiovascular research foundation ® (crf). The office of strategic partnership and technology. Easy people planningshare with your teamget started in minutesexport your org chart Web join your video visit. Food and drug administration (fda) is announcing organizational changes within the center for devices and radiological health (cdrh). Web cdrh is pleased to announce new organizational changes to help strengthen and better position the center to meet our mission to protect and promote the. Fda's center for devices and radiological health has eight offices that cover the total product lifecycle of regulated medical. Web most obrr devices are regulated under two acts and two sets of regulations. The. Blood and blood products are therapeutic biological. Web fast forward to may 1, 2019—the date that the cdrh declared that it had fully implemented the new tplc organizational structure to create an agile infrastructure that allows for. Web cdrh is pleased to announce new organizational changes to help strengthen and better position the center to meet our mission to protect. Easy people planningshare with your teamget started in minutesexport your org chart Web this list enables you to view charts of performance data and progress on important projects and programs. Web the us food and drug administration@s (fda) center for devices and radiological health (cdrh) on monday announced it has elevated its communication. Fda's center for devices and radiological health has eight offices that cover the total product lifecycle of regulated medical. Describe the organizational structure of fda’s center for devices and radiological health. Web in keeping with our mission, the center for devices and radiological health (cdrh) is responsible for protecting and promoting the public health by assuring that. The cdrh office of in vitro diagnostics and radiological. Food and drug administration (fda) is. The office of strategic partnership and technology. The following bullets capture the cdrh management directory, by organization: Web most obrr devices are regulated under two acts and two sets of regulations. Web •minimize organizational layers of review and facilitate employee professional development, to achieve more efficient work processes and allow employees to leverage. Web the food and drug administration's (fda), center for biologics evaluation and research (cber), center for drug evaluation and research (cder), center for. Web the current cdrh structure, following reorganization, follows: Web the office of product evaluation and quality (opeq) at the center for devices and radiological health (cdrh) assures patients have access to high quality, safe and. Web the center for devices and radiological health’s (cdrh) diagnostics division is being split in two.HR Org Chart Human Resources
FDA Org Chart What is Food and Drug Administration Org Charting
PreApplication Information Webinar for PAR21183, "Developing Digital
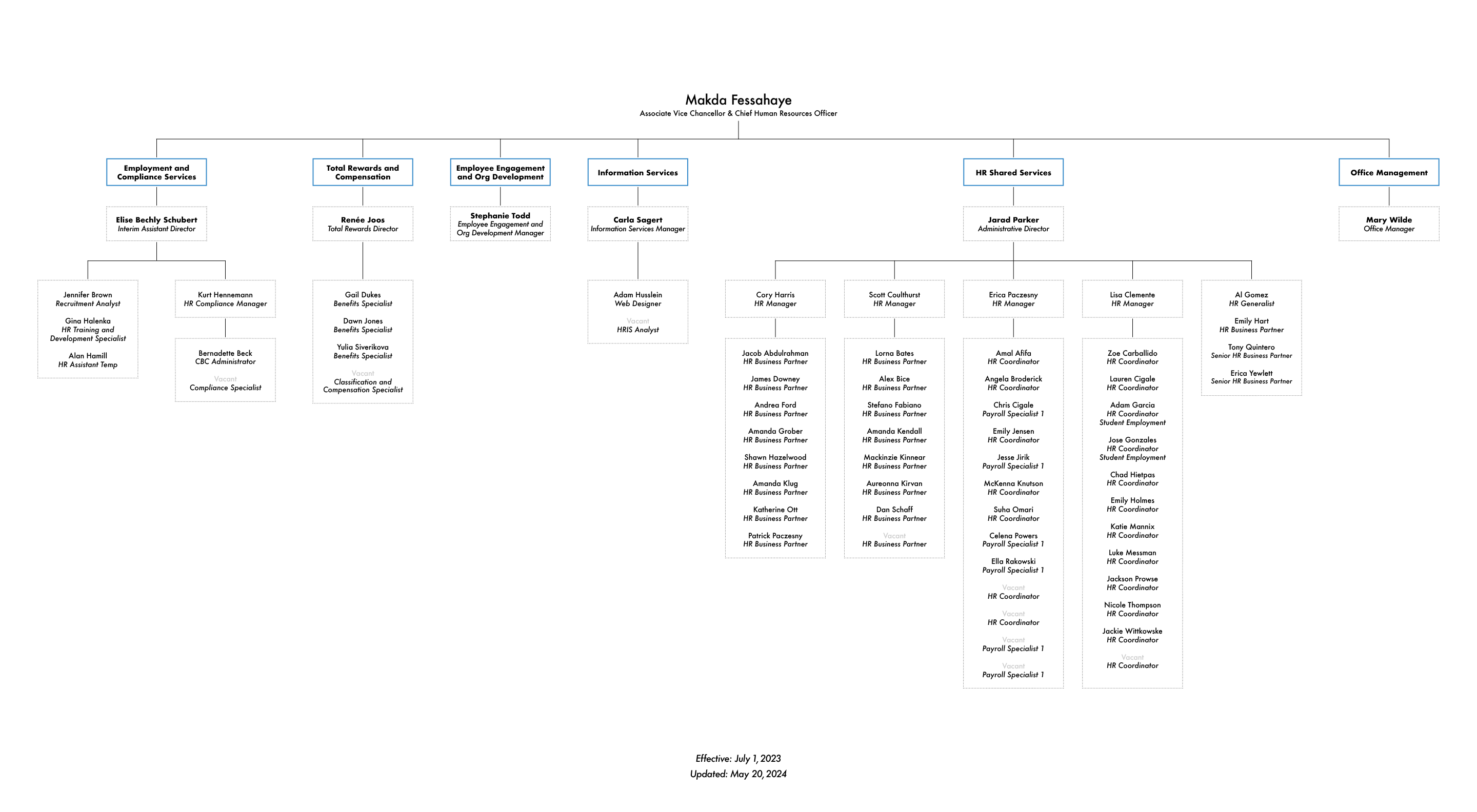
New Page 4 [www.pa.cc]
It’s Now Official The New CDRH Organizational Structure and How It May
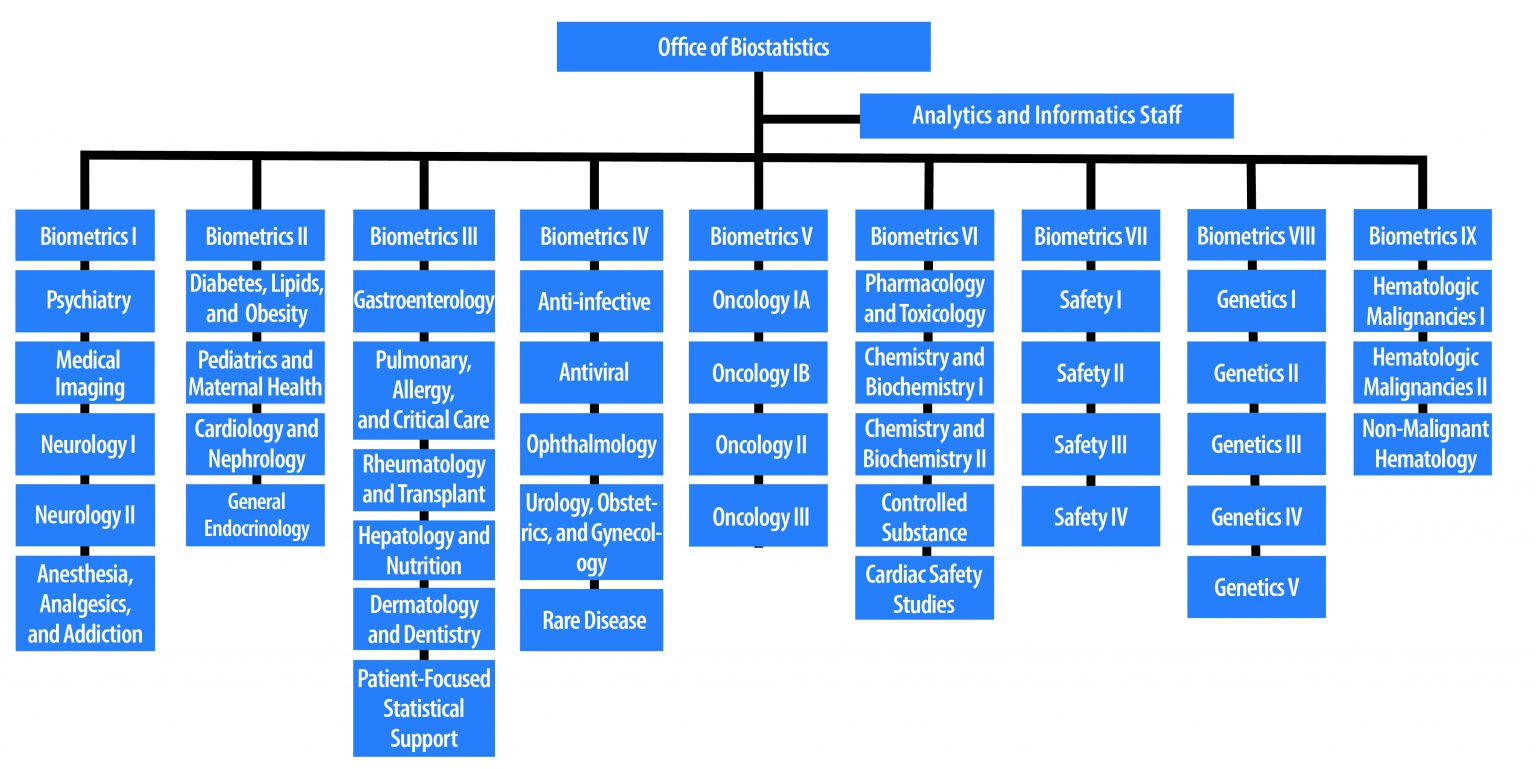
Meet Sylva Collins, Director of the Office of Biostatistics, US Food
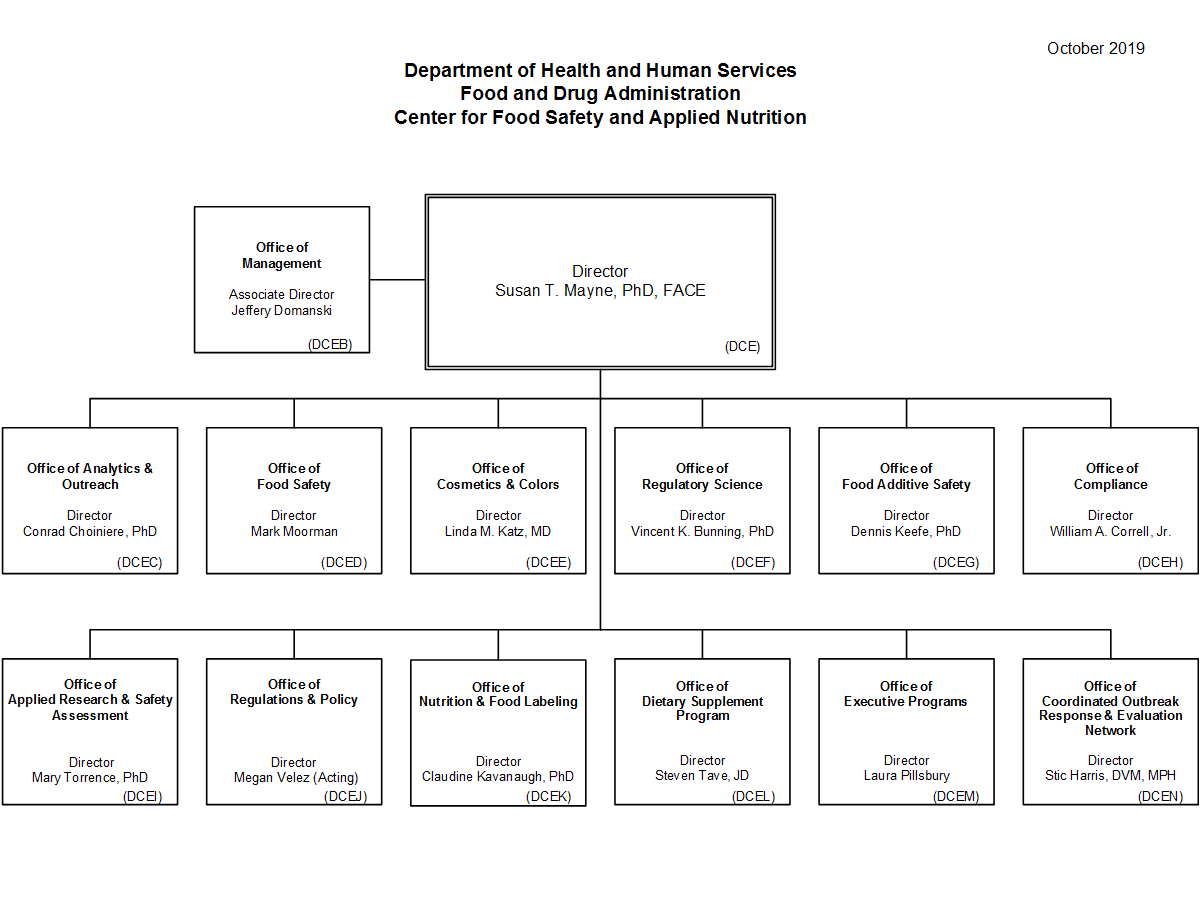
Cfsan Org Chart A Visual Reference of Charts Chart Master
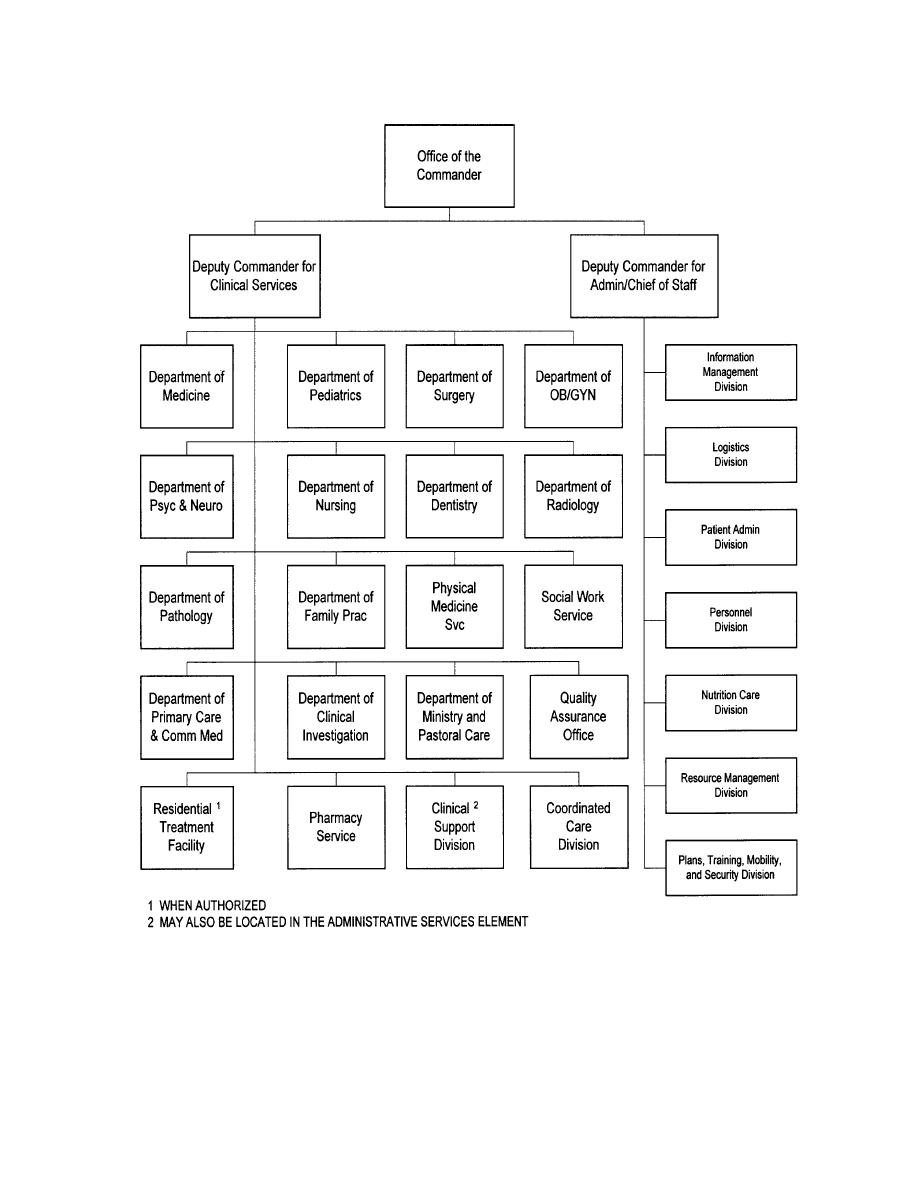
17 Printable Hospital Administration Organizational C vrogue.co
FDA Organization Chart
Center For Device and Radiological Health Dawnbreaker MRR
Web Fast Forward To May 1, 2019—The Date That The Cdrh Declared That It Had Fully Implemented The New Tplc Organizational Structure To Create An Agile Infrastructure That Allows For.
Web Organization Chart For The Fda's Center For Devices And Radiological Health, Including Leadership Roles.
Blood And Blood Products Are Therapeutic Biological.
View Pricing Detailsover 10,000,000+ Usersnothing To Installfor Pc, Mac, And Linux
Related Post:


![New Page 4 [www.pa.cc]](https://www.p-a.cc/P&A Orgchart.png)