Acid Base Chart
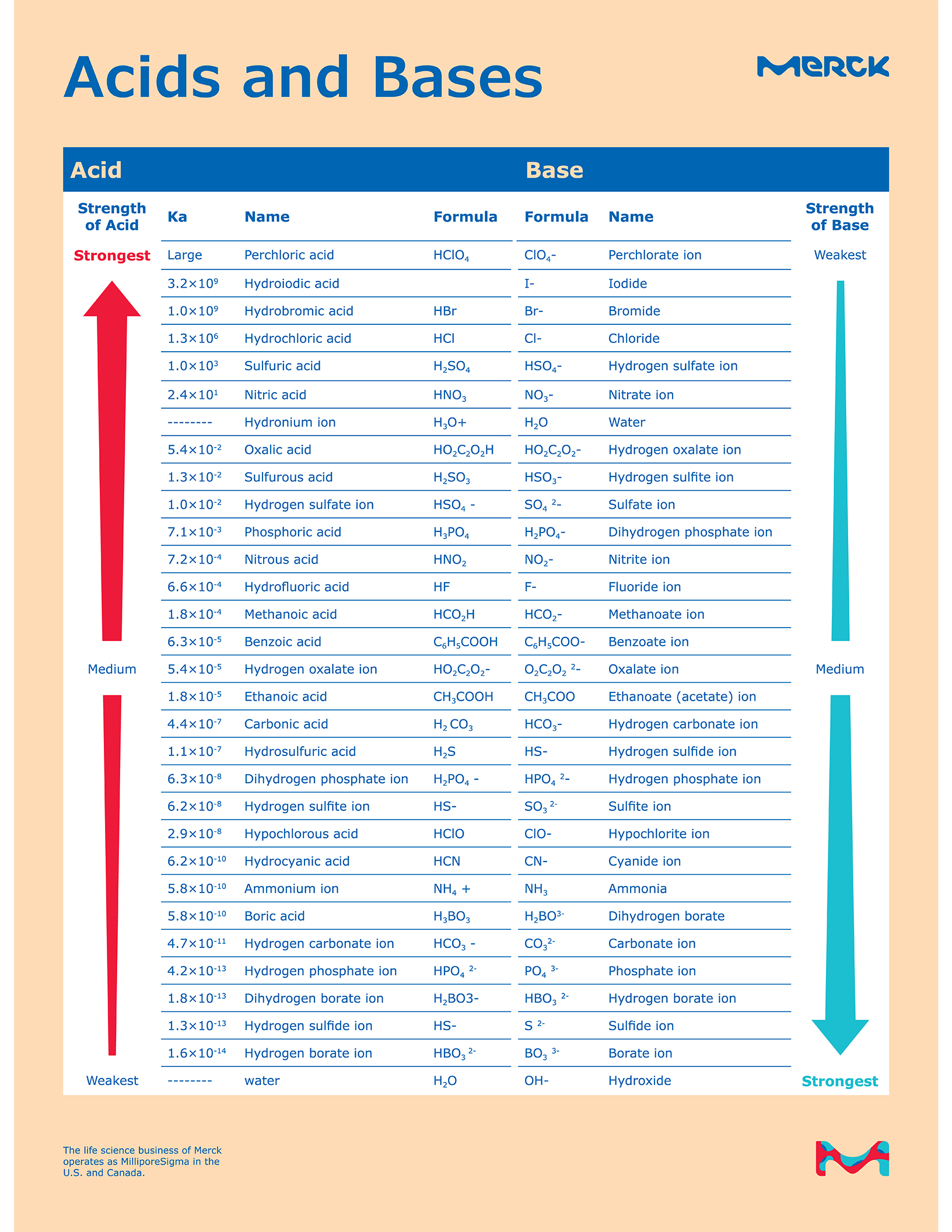
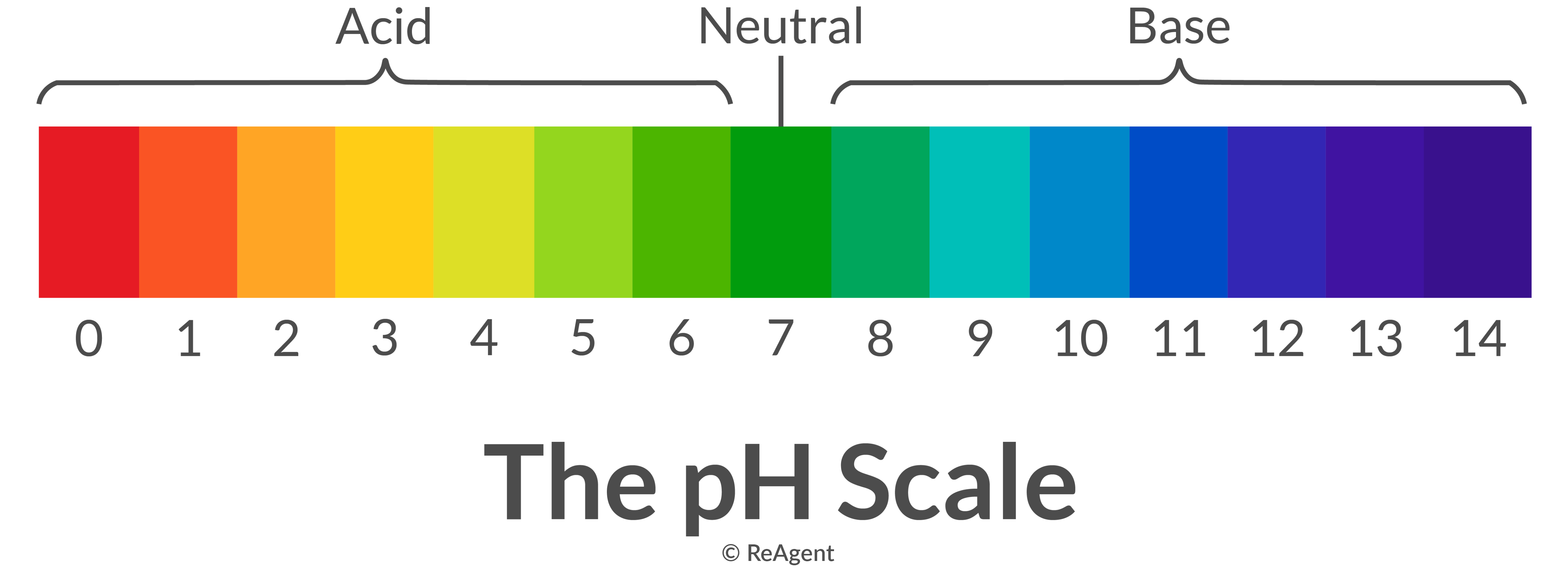
Acid Base Chart - Let’s start off with the ph value. Perchloric (hclo4), hydroiodic (hi), hydrobromic (hbr), hydrochloric (hcl), sulfuric (h2so4), nitric (hno3), and chloric. Chart or notebook size available. Web this is a list of the strong acids and strong bases. A ph less than 7 indicates acidity, while a ph greater than 7 indicates alkalinity. The strong acids ionize completely in water to yield or or more protons per acid molecule. Web the seven common strong acids listed from strongest to weakest are: Web in this tutorial, you will learn about the distinctive properties between acids and bases, what defines an acid vs base, and also what is an amphoteric compound. Solutions are classified as acidic or basic based on their hydrogen ion concentration relative to pure water. (select option to see volume pricing availability) 8½ × 11, pad of 30 (ap7229) $17.50. Let’s start off with the ph value. Web use this acids and bases chart to find the relative strength of the most common acids and bases. The strong bases are listed at the bottom right of the table and get weaker as we move to the top of the table. (select option to see volume pricing availability) 8½ × 11, pad of 30 (ap7229) $17.50. Web rules and resources. A ph less than 7 indicates acidity, while a ph greater than 7 indicates alkalinity. Web this is a list of the strong acids and strong bases. The ph scale, ranging from 0 to 14, measures the acidity or alkalinity (basicity) of a solution. Web acid with values less than one are considered weak. Chart or notebook size available. A ph less than 7 indicates acidity, while a ph greater than 7 indicates alkalinity. Web use this acids and bases chart to find the relative strength of the most common acids and bases. Web rules and resources. Chart or notebook size available. The strong acids ionize completely in water to yield or or more protons per acid molecule. Web use this acids and bases chart to find the relative strength of the most common acids and bases. Web in this tutorial, you will learn about the distinctive properties between acids and bases, what defines an acid vs base, and also what is an amphoteric compound. Acidic solutions have a higher h + concentration than water (greater than 1. 48 × 32, each (ap7042) $36.59. Let’s start off with the ph value. Chart or notebook size available. Pure water is an example of a substance with a neutral ph. Web this is a list of the strong acids and strong bases. Web acid with values less than one are considered weak. The ph scale, ranging from 0 to 14, measures the acidity or alkalinity (basicity) of a solution. Pure water is an example of a substance with a neutral ph. Web in this tutorial, you will learn about the distinctive properties between acids and bases, what defines an acid vs base,. The strong acids ionize completely in water to yield or or more protons per acid molecule. Web this is a list of the strong acids and strong bases. Pure water is an example of a substance with a neutral ph. 48 × 32, each (ap7042) $36.59. Web use this acids and bases chart to find the relative strength of the. Web acid with values less than one are considered weak. 48 × 32, each (ap7042) $36.59. Solutions are classified as acidic or basic based on their hydrogen ion concentration relative to pure water. (select option to see volume pricing availability) 8½ × 11, pad of 30 (ap7229) $17.50. Acidic solutions have a higher h + concentration than water (greater than. The strong bases are listed at the bottom right of the table and get weaker as we move to the top of the table. Pure water is an example of a substance with a neutral ph. Web this is a list of the strong acids and strong bases. There aren’t very many, so it’s a good idea to memorize them,. Chart or notebook size available. There aren’t very many, so it’s a good idea to memorize them, if you can. 48 × 32, each (ap7042) $36.59. A ph less than 7 indicates acidity, while a ph greater than 7 indicates alkalinity. A ph of 7 is neutral. Pure water is an example of a substance with a neutral ph. Let’s start off with the ph value. There aren’t very many, so it’s a good idea to memorize them, if you can. Web use this acids and bases chart to find the relative strength of the most common acids and bases. (select option to see volume pricing availability). A ph of 7 is neutral. 48 × 32, each (ap7042) $36.59. The strong bases are listed at the bottom right of the table and get weaker as we move to the top of the table. Pure water is an example of a substance with a neutral ph. Web acid with values less than one are considered weak. Web in this tutorial, you will learn about the distinctive properties between acids and bases, what defines an acid vs base, and also what is an amphoteric compound. Let’s start off with the ph value. 48 × 32, each (ap7042) $36.59. (select option to see volume pricing availability) 8½ × 11, pad of 30 (ap7229) $17.50. The ph scale, ranging from 0 to 14, measures the acidity or alkalinity (basicity) of a solution. Web acid with values less than one are considered weak. A ph of 7 is neutral. Perchloric (hclo4), hydroiodic (hi), hydrobromic (hbr), hydrochloric (hcl), sulfuric (h2so4), nitric (hno3), and chloric. Web this is a list of the strong acids and strong bases. There aren’t very many, so it’s a good idea to memorize them, if you can. Web rules and resources. Chart or notebook size available. Acidic solutions have a higher h + concentration than water (greater than 1 × 10 − 7 m), while basic (alkaline) solutions have a lower h + concentration (less than 1 × 10 − 7 m). Web the seven common strong acids listed from strongest to weakest are: A ph less than 7 indicates acidity, while a ph greater than 7 indicates alkalinity. Solutions are classified as acidic or basic based on their hydrogen ion concentration relative to pure water.Interpretation Acid Base Tutorial
Acid and Base Chart — Table of Acids & Bases
Water, Acids, and Bases CK12 Foundation
Acids and Bases
List of Strong Acids & Bases in Order StudyPK
Back to Basics Acids, Bases & the pH Scale Precision Laboratories
What’s The Reaction Between An Acid And A Base?
pH Of Acids And Bases Calculate pH Value Chemistry Byju's
AcidBase Strength Charts for Chemistry
16.5 Strong Acids and Bases Chemistry LibreTexts
The Strong Acids Ionize Completely In Water To Yield Or Or More Protons Per Acid Molecule.
Pure Water Is An Example Of A Substance With A Neutral Ph.
Web Use This Acids And Bases Chart To Find The Relative Strength Of The Most Common Acids And Bases.
The Strong Bases Are Listed At The Bottom Right Of The Table And Get Weaker As We Move To The Top Of The Table.
Related Post: